BACKGROUND and MEANING of most ESSENTIAL FIRE ASSAY TERMS – download
BLANK ASSAY
The best reagents obtainable for assaying contain some gold and silver. As a rule, the amount is so small that it may be neglected, but this point should always be determined by “blank” assays of the reagents. Each new lot of reagents should be tested by a blank assay, made with assay silica that is treated as a quartz ore (Shepard. 228).
The fluxes and other reagents commonly employed in dry assaying do not contain silver or gold, but if they are left exposed in the laboratory they sometimes become accidentally (and perhaps purposely) contaminated or “salted” with material containing gold and silver values.
A blank assay (that is, a charge in which the ore is omitted) of the reagents will readily determine whether any precious metal is present. (Smith p. 145 -146)
BLICK / BLINK / BIRGHTENING / FLASH / SCINTILLATION”
After cupellation, it is key to slowly cool the beads. Otherwise Sprouting or spitting may occur due to the fact that molten silver is able to dissolve greats amounts of oxygen, and violently releasing it when it solidifies again. When spitting occurs the assays needs to be rejected because the spitting phenomena is nearly always accompanied by a loss of precious metals.
FEATHERS
Feathers are sublimed crystals of solid litharge that deposit on the rim of the cupel, which is invariably cooler than the molten lead on the cupel. For Magnesia Cupels, the abundance of Feathers is a sign that the assay temperature is near the freezing point.
As cupellation proceeds and the lead button becomes smaller, concentric rings of feathers deposit within the original ring., However at all times the temperature should be high enough so that there is a clear area between the button and the feathers, otherwise there is danger that a pool of litharge will form immediately around the button, which will instantly prevent further absorption of litharge by the cupel, and the litharge will soon completely cover the lead and solidify, which will cause the button to freeze.
Even if the assayer fails to note the encroachment of feathers toward the button, or the decrease in color temperature of the lead, the onset of freezing is plainly evident in the oily appearance of the ring of molten litharge at the outer periphery of the button. If this happens we have to raise temperature immediately to avoid freezing.
Individual cupels that show signs of incipient freezing may be saved by placing a hot cupel or brick near or over them. (Shepard p. 59)
FREEZING OF ASSAY
During cupellation, if the temperature becomes too low for the cupel to absorb the litharge, the crystals begin to form all around and close to the lead in the cupel. Soon a pool of molten litharge forms all around the annular space between the lead and the cupel. If the temperature of the cupel is not quickly raised, this pool increases in size and soon entirely covers the lead and then solidifies. When this occurs the button has “frozen,” although the lead itself may be liquid underneath.
Frozen assays are rejected as the results obtained from them, by again bringing to a driving temperature, are usually low. It is possible to arrest the freezing if is noticed at the start, and if the temperature of the cupel is quickly raised in some way, i. e., by taking away the coolers, closing the door to the muffle, opening the draft, putting a hot piece of cokei in front of the cupel, etc. (Bugbee p. 93) All assayers agree that the best results are obtained with a hot start, a cold drive, and a higher heat again at the finish. (Bugbee p. 98)
MATTE – SPEISS – REGULUS
MATTE is an artificial sulphide of one or more of the metals, formed in the dry way. In assaying it is most often encountered in the niter fusion of sulphide ores when the charge is too acid. It is found lying just above the lead button. It is usually blue-gray in color, approaching galena in composition and very brittle. It may form a layer of considerable thickness, or may appear simply as a granular coating on the upper surface of the lead button. This matte always carries some of the gold and silver and, as it is brittle, it is usually broken off and lost in the slag in the cleaning of the lead button. The student should examine the lead button as soon as it is broken from the slag and if any matte is found, he may be certain that his charge or furnace manipulations are wrong.
SPEISS is an artificial, metallic arsenide or antimonide formed in smelting operations. As obtained in the fire assay, it is usually an arsenide of iron approaching the composition of Fe5As. Occasionally the iron may be replaced by nickel or cobalt. The antimony speiss is very rare. In assaying, speiss is obtained when the iron method is used on ores containing arsenic. It is a hard, fairly tough, tin-white substance found directly on top of the lead and usually adhering tenaciously to it.
If only a small amount of arsenic is present in the ore, the speiss will appear as a little button lying on top of the lead; if much arsenic is present, the speiss will form a layer entirely covering the lead. It carries some gold and silver. If only a gram or so in weight, it may be put into the cupelwith the lead and will be oxidized there, giving up its precious metal values to the lead bath. A large amount of speiss is very hard to deal with as it is difficult to scorify. The best way is to assay again, by some other method. (Bugbee p. 15)
One way to avoid the formation of speiss and matte is to charge a sufficient excess of litharge and soda to keep antimony, arsenic and sulfur oxidised,
OPENING AND DRIVING OF ASSAY
Once, we have carefully placed the lead button in the cupels and in the muffle, if we heat the cupels thoroughly, the lead will melt at once and become covered with a dark scum. If the temperature of the muffle is correct this will disappear in the course of a minute or two when the molten lead will become bright. The assays are then “opened up or “uncovered.” This signifies that the lead has begun to oxidize rapidly, raising the temperature of the molten alloy considerably above that of its surroundings, whence it appears bright. It assumes a convex surface, and molten patches of litharge passing down over this surface give it a lustrous appearance. It is “driving.” (Bugbee p. 92)
“PLAY OF COLOURS” – “BRIGHTENING”
The visual phenomena that appear near, and at the finish of cupellation are of considerable aid to the assayer.
Since both silver and gold have a higher surface tension than lead the button becomes more rounded as the percentage of lead decreases; this effect becomes pronounced as the proportion of precious metals increases beyond 50%.
When the cupellation of large beads approaches completion, oily-appearing drops of litharge can be seen to collect on the surface of the bead. These particles appear to move, and the movement becomes increasingly more rapid until just before the finish, when the molten lithargeforms a thin film of variable thickness and creates interference colors.
The rainbow color bands move swiftly over the surface of the button and give the illusion that the button is revolving about a shifting axis. This is known as the “play of colors” and is strikingly developed with large beads.
When the last trace of lead has been removed from the bead the play of colors disappears and the bead becomes dull for a brief period, after which it acquires a normal metallic luster. This change of luster is known as “brightening” and is not always observable. (p. 62)
“SPIT” – “SPITTING” at beginning of cupellation / fire assaying
Before cupellation the set of cupels should be charged into the furnace and heated at 850 to 900°C for 10 min., with the draft slightly open to provide an oxidizing atmosphere. This will drive off free and combined water,
organic matter, carbon dioxide, and other volatile constituents that would otherwise rise through the lead during cupellation and cause the ejection of particles of lead.
This phenomenon is known as “spitting” and is a source of loss to the assay in question, as well as the cause of salting other samples into which the globules of lead may fall. (Shepard p. 56)
Ores that decrepitate violently are troublesome in scorification because of “spitting,” which is the violent projection of small particles of lead from the scorifier.
… Place the scorifier into a muffle at 500 to 600°C, close the door and muffle draft, and heat for 2 or 3 min. until the lead is melted and danger of decrepitation or spitting is past. (Shepard p. 166)
“SPIT” – “SPITTING” – “SPROUT” – “SPROUTING” – “SPURTING”- “VEGETATION” at the end of cupellation
Liquid silver has the peculiarity of dissolving a large volume of oxygen (22 times its volume in oxigen) , which is expelled upon solidification. The solidification of large beadsstarts at the surface. When the center of silver beads solidifies, the oxygen is sometimes expelled violently, spewing forth some of the interior silver and forming a cauliflower-like growth on the bead. This mechanism is known as “sprouting.”
Sprouted beads should be rejected if there is any reason to believe that, any particles of metal have been lost.
To avoid sprouting, all large beads should be cooled slowly, either by leaving them in the, furnace after the blick, and until certain that, they have cooled sufficiently, or by moving them to a slightly cooler zone in the furnace and then covering them with a very hot cupel before they have completely solidified.
The hot cupel melts the outer crust and allows the bead to solidify slowly, so that the oxygen can escape from the interior without violence. When the bead contains more than one-third of its weight in gold, it does not sprout; hence beads known to be of this composition may be removed from the furnace as rapidly as desired.
Sprouting of silver beads is considered a sign of purity, but this indication is of no practical value to the assayer, as sprouting should always be avoided. (Shepard p. 63)
Bugbee states that as little as 0.004% of rhodium in silver beads causes a distinct crystallization, which is more apparent at 0.01% Rh, and that 0.03% Rh causes unavoidable sprouting of silver beads. (Shepard p. 71)
“SURCHARGE” – “CHECK” – “PROOF” – “STANDARD”
Surcharge — Net Losses and Gains — The net sum of the (…) losses and gains incurred in the various operations is called the “surcharge” since, in the assay of high-grade bullion, the silver retained is usually in excess of the gold lost, so that the cornet will weigh more than the gold originally present in the assay piece; if the reverse is the case, the result is considered by some assayers to be less accurate. When there is a gain in weight it is referred to as a “plus surcharge“, while a loss in weight is designated a “minus surcharge“.
With material rich in gold, the silver retained more than compensates for the loss, and, as just stated, the surcharge is positive; but with a decrease in the proportion of gold the loss is greater and the surcharge is negative.
Thus the surcharge will usually amount to about 0 (nil) for bullion of about 700 to 800 fine; above that there will be a “plus surcharge” and below that a “minus surcharge“. The surcharge is reported in parts per 1000 in the same way as the fineness of bullion. (Smith p. 276)
“INQUART” – “INQUARTATION”
Inquartation. – When the bead contains too little silver to part, it is necessary to alloy it with more silver. This process is called inquartation. It originated from the custom of the old assayers of adding silver until the gold was one-quarter of the whole. They considered a ratio of 3 parts of silver to 1 of gold to be necessary for parting. At present, in assaying gold bullion, a ratio of only 2 or 2¼ parts of silver to 1 of gold is used, mainly to avoid all danger of the gold breaking up in the boiling acid.
In this case some little silver remains undissolved, even though the alloy is rolled out to about 0.01″ in thickness. (Bugbee p. 121)
Many assayers, when working for both gold and silver and suspecting an ore to be deficient in silver, add silver to the crucible or to the leadbutton before cupeling, part directly and then run separate assays to determine the silver in the ore. (Bugbee p. 122)
MICROCUPELLATION
Microcupellation consists of doing the cupellation process but with a much smaller sample.
The sample size for cupellation is tipically 250mg, but with microcupellation it is only about 10 mg. This allows not to “destroy” the jewelry piece to analyse as the sample can sometimes also be taken from hidden places of the jewelry article.
An additional advantage of microcupellation is that it takes less time than conventional cupellation.
CUPELLATION
The cupel is a shallow, porous dish made of bone-ash, Portland cement, magnesia or other refractory and non-corrosive material. (Bugbee p. 89)
Cupels should not crack when heated in the muffle and should be so strong that they will not break when handled with the tongs. Good cupels give, a slight metallic ring when struck together after air-drying. It is best to heat cupels slowly in the muffle as this lessens the chance of their cracking.
A good cupel should be perfectly smooth on the inside, and of the right porosity. If it is too dense, the time of cupellation is prolonged and the temperature of cupellation has to be higher, thus increasing, the loss of silver. If the cupel is too porous it is said that there is danger of a greater loss, due to the ease with which small particles of alloy can pass into the cupel. The bowl of the cupel should be made to hold a weight of lead equal to the weight of the cupel.
The shape of the cupel seems to influence the loss of precious metals. A flat, shallow one exposes a greater surface to oxidation and allows of faster cupellation; it also gives a greater surface of contact between alloy and cupel, and as far as losses are due to direct absorption of alloy, it will of course increase these.
The writer, using the same bone-ash and cupel machine, and changing only the shape of the cupel, has found shallow cupels to give a much higher loss of silver. In doing this work it was found harder to obtain crystals of litharge with the shallow cupel without freezing, and it was very evident that a higher cupellation temperature was required for the shallow cupel. The reason for this is that in the case of the shallow cupel the molten alloy is more directly exposed to the current of air passing through the muffle, and consequently a higher muffle temperature has to be maintained to prevent freezing. T. K. Rose* also prefers deep cupels on account of smaller losses. French found shallow cupels less satisfactory on account of sprouting. (Bugbee p. 92 – 93)
Magnesia cupels are very hard, which is an advantage in that they do not suffer so much breakage in shipment. They are always factory-made and are decidedly more expensive than bone-ash cupels, which may be home-made. Certain brands of magnesia cupels give an apparently lower loss of silver in cupeling than can be obtained with bone-ash cupels but it is a question how much of this is real and how much due to an increase in the amount of impurities retained in the silver beads.
Magnesia cupels behave quite differently from ordinary bone-ash cupels, and the assayer who is accustomed to bone-ash cupels will have to learn cupeling over again when he starts using those made of magnesite. This difference in behavior is due mainly to the different thermal properties of the two materials. Both the specific heat and the conductivity of magnesite are decidedly greater than those of bone-ash, so that with cupels of both kinds running side by side, the lead on the magnesia cupel is comparatively dull while that on bone-ash is very bright. This is due to the greater conductivity of magnesite, which allows a more rapid dispersion of the heat of oxidation of the lead, with the result that magnesia cupels require a higher muffle temperature than do bone-ash cupels. An especially high finishing temperature is required for magnesite cupels, to insure the elimination of the last 1 or 2% of lead. A bone-ash cupel will finish in a muffle, the temperature of which is sufficient to cause uncovering, but this is not true of the magnesia cupel, because in this case the heat of oxidation of the lead is diffused too rapidly and is not conserved to help out at the finish.
Magnesia cupels absorb about two-thirds of their own weight of litharge, those of cement about three-fourths of their weight of litharge. (Bugbee p. 116 – 117)
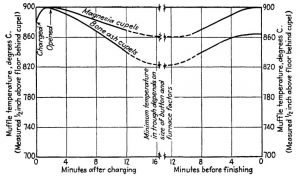
- TEMPERATURE CYCLE DURING CUPELLATION
Summary of Cupellation–temperature Cycle. — Characteristic temperature cycles in cupellation are shown on Fig. 8, based on measurements taken with a pyrometer ½” above the muffle floor just behind the front row of cupels. The curves apply particularly to buttons from 20 to 25 g in weight and with beads weighing 50 mg or more, cupeled with feathers.
The indicated temperatures are subject to corrections based on the furnace draft, heating and cooling lag, and the pyrometer position.
The minimum temperature allowable during the driving depends also upon the type of cupel used and the size of the button and bead. Large buttons that are cupeled rapidly in bone-ash cupels with the ordinary shallow cup frequently permit minimum muffle temperatures as low as 790°C and will finish at 820°C if the beads are small.
Magnesia cupels under similar conditions require minima of 830°C in the driving trough, and 840° to 850°C to finish.
The formation of feathers of litharge can be observed readily with bone-ash or bone-ash-cement cupels and serves to indicate proper driving temperature, but when copious feathers form on magnesia cupels the temperature is dangerously near the freezing point.
Finishing temperatures greatly in excess of 900°C, as measured in the manner indicated above, should be avoided in all cases, as higher temperatures cause greatly increased losses of gold and silver with all types of cupels and all variations in button and bead weight. (Shepard p. 66 – 67)
Figure 8: Cupellation Temperature Cycle
ANALYTICAL METHODS FOR DETERMINATION OF GOLD ( Articles & Papers )
- Assaying of Gold Jewellery – Review of fire assay methods
- “Assaying of Gold Jewel. – Choice of Technique” (“Gold Technology” Magazine (WGC), nº 32, 2001) – Dr. C. W. Corti
- “Analysis of Carat Gold” (“Gold Technology” Magazine (WGC), nº 22, Jul. 1997) – M. Brill
- “Assaying and Hallmarking in London” (“Gold Technology” Magazine (WGC), nº 3, Jan 1991) – D. W. Evans
- “Assaying Gold in Switzerland” (“Gold Technology” Magazine (WGC), nº 3, Jan 1991) – Wälchi / Vuilleumier
- “Touchstone Testing of Precious Metals” (“Gold Technology” Magazine (WGC), nº 3, Jan 1991) – Wälchi / Vuilleumier
- “Analysis of Gold. A Review of Methods” (“Gold Bulletin” Journal” (WGC), Vol. 13, issue 1) – Roland S. Young
- “The Art & Science of a Precious Metal Lab.” (“Alchemist” Journal, (LBMA)., nº 48, LBMA) – David Court
- “Analytical Chemistry of Noble Metals” (Pure & Appl. Chem., Vol. 49) Dr. Jon Cl. van Loon
REFERENCE BOOKS – Free download ►Internet Archive ►(I.A.) ►Bewster Kahle
- “A Textbook on Fire Assay” – Edward E. Bugbee
- “Fire Assaying” – Orson C. Shepard & Waldemar F. Dietrich
- “A Manual on Fire Assaying” – Charles Herman Fulton
- “The Sampling and Assay of the Precious Metals” – Ernest Alfred Smith
- “Metallurgy of Gold” – Thomas K. Rose
- “The Precious Metals: comprising Gold, Silver and Platinum” – Thomas K. Rose
- “A Text Book of Assaying” – C. Beringer & J. J. Beringer